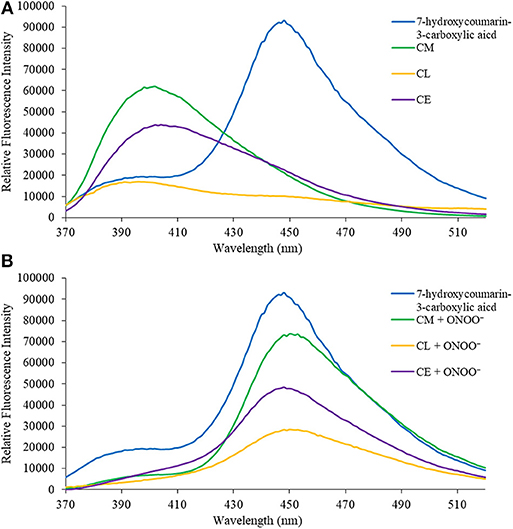
Figure 2 from Understanding the fate of peroxynitrite in plant cells--from physiology to pathophysiology.
€ 16.50 · 4.6 (391) · Auf Lager

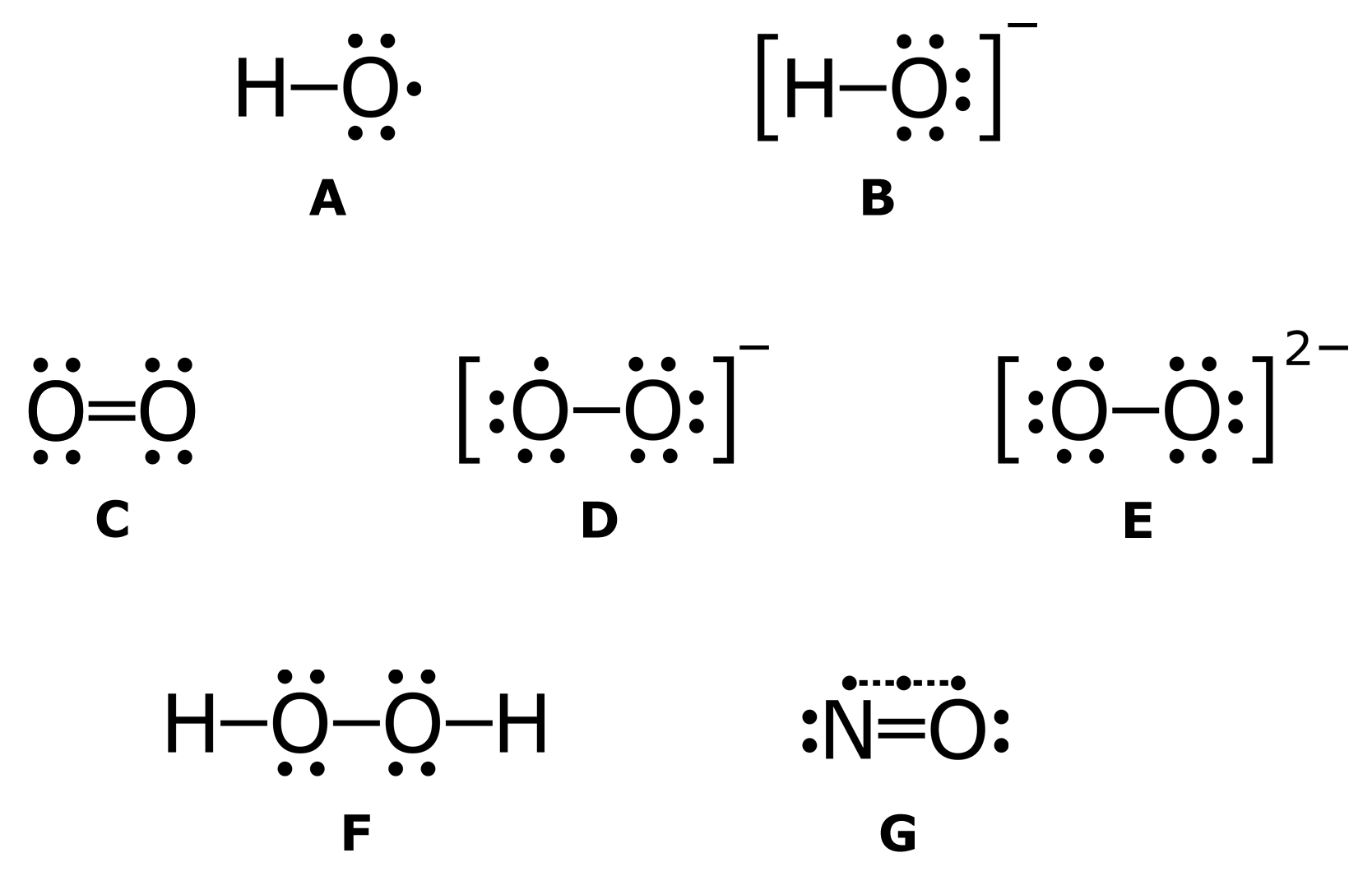
Fig. 2. A hypothetical regulatory mechanisms of ONOO action in plant cells. In the biological milieu the peroxynitrite anion (ONOO ) is in equilibrium with peroxynitrous acid (ONOOH; pKa = 6.8). The reaction of ONOO with carbon dioxide leads to the formation of carbonate (CO 3 ) and nitrogen dioxide ( NO2) radicals. Alternatively, ONOOH can undergo homolytic fission to generate one-electron oxidants, hydroxyl OH and NO2 radicals. ONOOH readily crosses lipid bilayers and its decomposition to OH and NO2 radicals seems to become relevant in hydrophobic phases to initiate lipid peroxidation and lipid and protein nitration processes. If ONOO combines with SH-containing molecules (X-SH), it might be converted to S-nitroso compounds, e.g. S-nitrosoglutathione. Nitration and the formation of S-nitroso compounds are proposed mechanisms, by which ONOO regulates NO-dependent signaling events. On the other hand, nucleotides within DNA and RNA can undergo nitration by ONOO forming 8-nitroguanine, which may impart pathological consequences (Corpas et al., 2009b). - "Understanding the fate of peroxynitrite in plant cells--from physiology to pathophysiology."
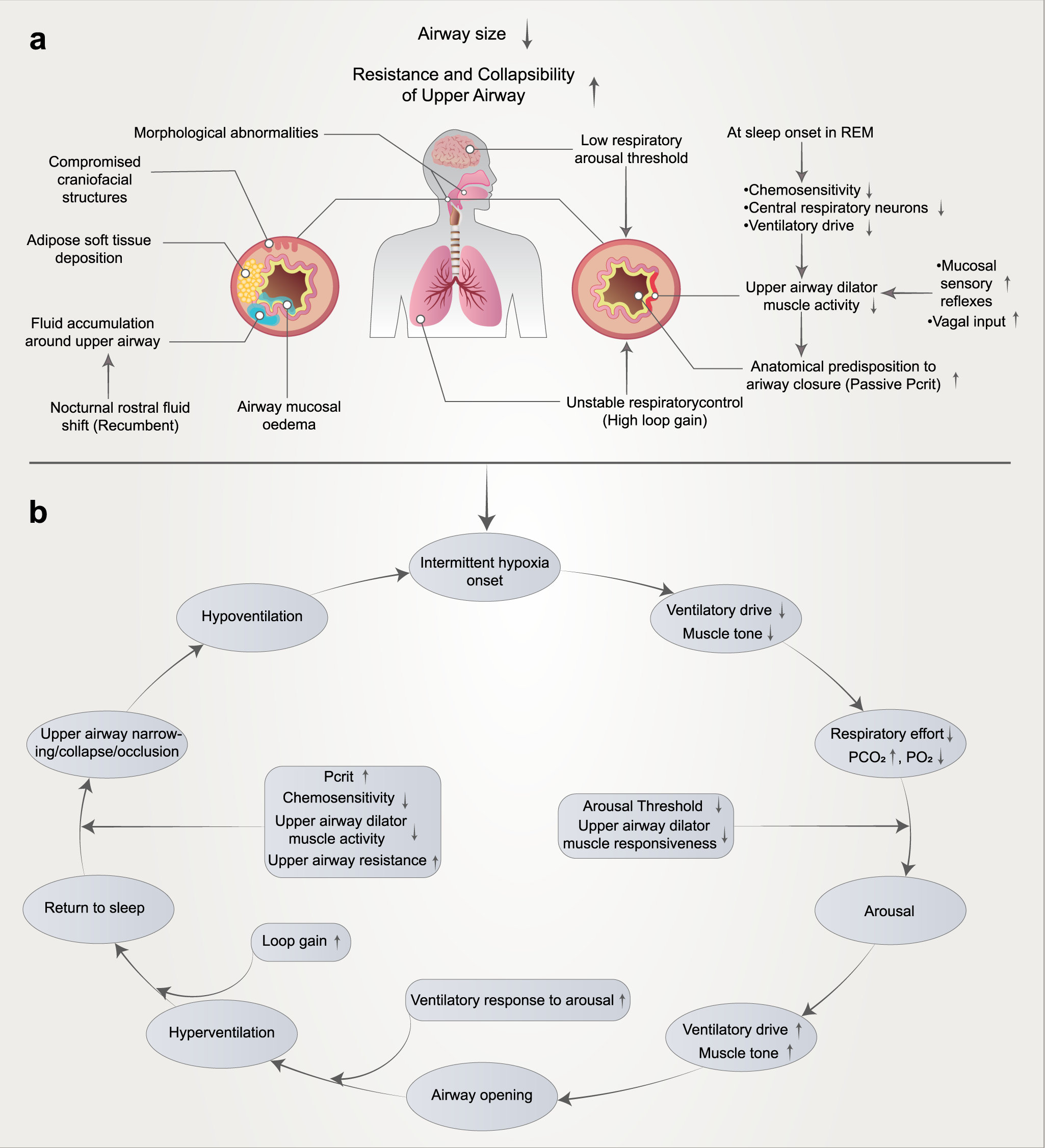
Pathophysiological mechanisms and therapeutic approaches in
Role of reactive oxygen (ROS) and reactive nitrogen (RNS) species
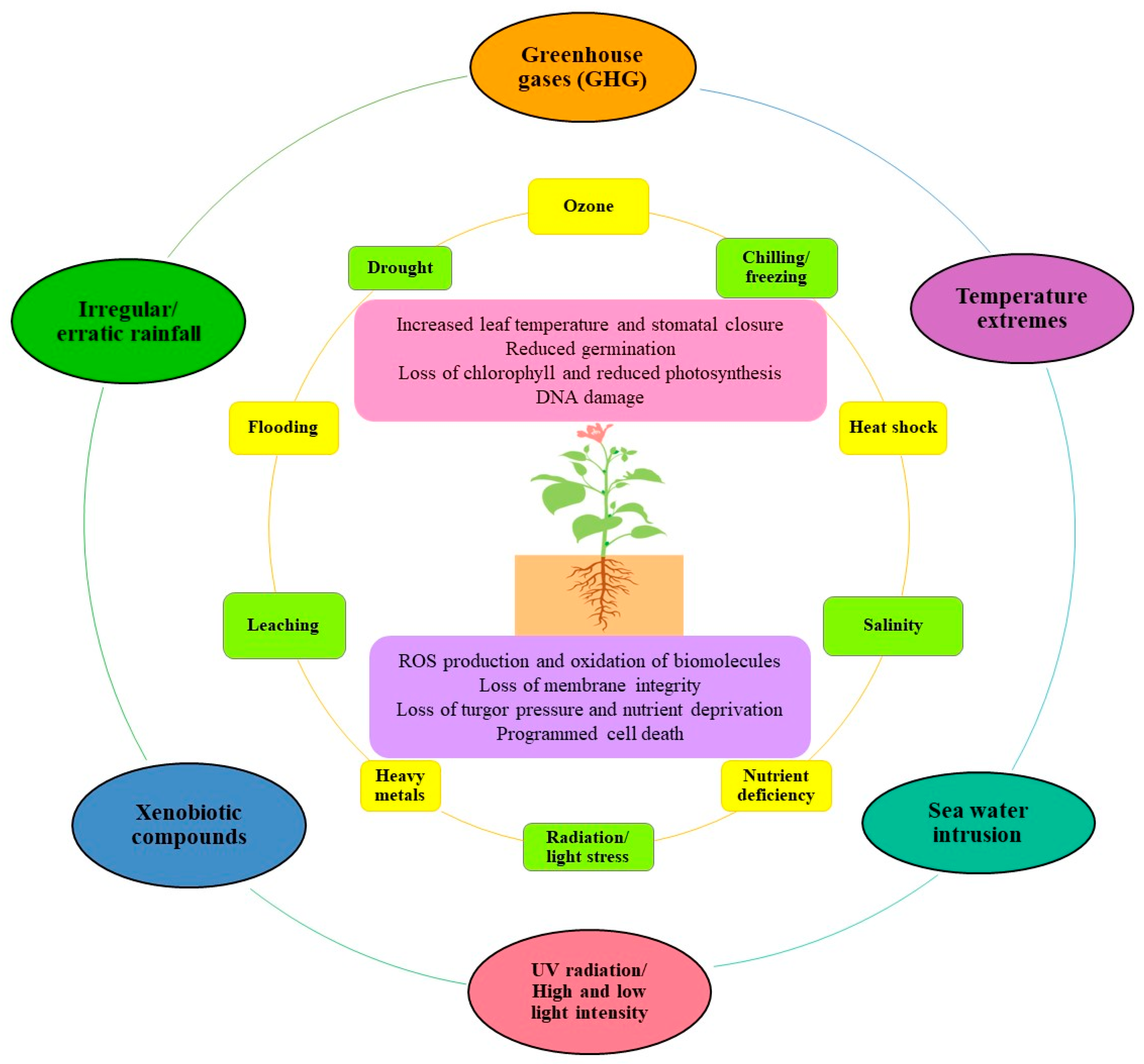
Antioxidants, Free Full-Text

Mitochondria in Control of Cell Fate

Overview of peroxynitrite reactivity with lipids. Å NO and

Figure 6 from Nitric oxide and peroxynitrite in health and disease

NOS inhibition reverses TLR2-induced chondrocyte dysfunction and

The nexus between redox state and intermediary metabolism

Full article: Peroxynitrite: cellular pathology and implications
Reactive oxygen species - Wikipedia

Reactive Oxygen Species: Physiological and Physiopathological

Journal of Cellular Physiology, Cell Biology Journal

Journal of Cellular Physiology, Cell Biology Journal

Neuroprotective Effect of NO-Delivery Dinitrosyl Iron Complexes